MiGrampa
Herb Hitman
- Joined
- May 18, 2023
- Messages
- 665
- Reaction score
- 2,832
Update:
After scraping the substrate with a fork and adding a casing, I finally got the KSSS to fruit. I'm glad I stuck with it. I'm seeing a lot of small pins starting to form.

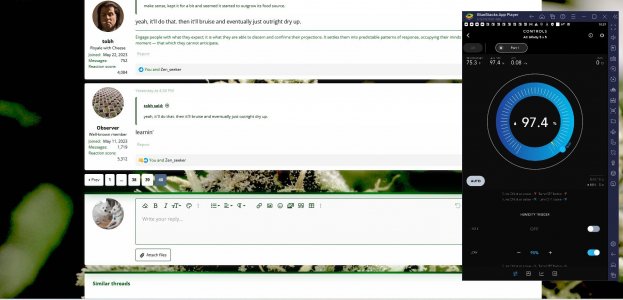
Also, using my AC Infinity UIS69 Pro controller and a small tent I already had, I've automated my mushroom grow. Top shelf is the KSSS and the middle shelf is the Pans Cyan. The controller is set to turn the humidifier on when humidity drops below 95%.
I'm still learning as I go so suggestions/tips are encouraged.

After scraping the substrate with a fork and adding a casing, I finally got the KSSS to fruit. I'm glad I stuck with it. I'm seeing a lot of small pins starting to form.

Also, using my AC Infinity UIS69 Pro controller and a small tent I already had, I've automated my mushroom grow. Top shelf is the KSSS and the middle shelf is the Pans Cyan. The controller is set to turn the humidifier on when humidity drops below 95%.
I'm still learning as I go so suggestions/tips are encouraged.



















